Medical Terminology Daily (MTD) is a blog sponsored by Clinical Anatomy Associates, Inc. as a service to the medical community. We post anatomical, medical or surgical terms, their meaning and usage, as well as biographical notes on anatomists, surgeons, and researchers through the ages. Be warned that some of the images used depict human anatomical specimens.
You are welcome to submit questions and suggestions using our "Contact Us" form. The information on this blog follows the terms on our "Privacy and Security Statement" and cannot be construed as medical guidance or instructions for treatment.
We have 835 guests online

Marcia Crocker Noyes
(1869 – 1946)
Further to my comment on old books and research that started with an interesting bookplate (Ex-Libris). I continued my research and found that the person in charge of the Osler library bookplate was a fascinating individual that today maybe a ghost in the MedChi library and building in Baltimore... This is certainly an article that can be called "A Moment in History"
Marcia Crocker Noyes was the librarian at The Maryland State Medical Society from 1896 to 1946 and was a founding member of the Medical Library Association.[1][2][3]
Sir William Osler, MD. a famous Johns Hopkins surgeon was a noted bibliophile and had a large personal collection of books on various topics. When he became the President of MedChi in 1896, he was dismayed at the condition of the library and knew that with the right person and some stewardship, it could become a significant collection. Sir William asked his friend, Dr. Bernard Steiner, a physician and President of the Enoch Pratt Free Library in Baltimore for suggestions of a librarian, and Dr. Steiner recommended Marcia Crocker Noyes. A native of New York, and a graduate of Hunter College, Marcia had moved to Baltimore for a lengthy visit with her sister, and took a “temporary” position at the Pratt Library, which turned into three years. Although she had no medical experience or background, she was enthusiastic, and most importantly, she was willing to move into the apartment provided for the librarian, who needed to be available 24 hours a day.
The image in this article is Ms. Noyes on her first year on the job. Marcia developed a book classification system for medical books, based on the Index Medicus, and called it the Classification for Medical Literature. The system uses the alphabet with capital letters for the major divisions of medicine and lower-case ones for the sub-sections. The system was used for many years, but it's now dated and the Faculty's original shelving scheme was never changed. The card catalogs still reflect her classification and many of the cards are written in Marcia's back-slanting handwriting.
Marcia knew enough to ask the Faculty's members about medical questions, terminology and literature. She gradually won over the predominantly male membership and they became her greatest allies; Sir William at the start, and then for nearly 40 years, Dr. John Ruhräh, a wealthy pediatrician with no immediate family of his own. She made a point of attending almost every Faculty function, and in 1904, under guidelines from the American Medical Association, Marcia was made the Faculty Secretary. For much of her first 10 years, she was the Faculty's only full-time employee, only being assisted by Mr. Caution, the Faculty's janitor. Later in life Marcia would say that she hired him because of his name!
Within ten years, the library had outgrown its space, and plans, spearheaded by Marcia and Sir William before his move to Oxford, were made to build a headquarters building, mainly to house the library's growing collection of medical books and journals.
Marcia was instrumental in the design and building of the new headquarters. She travelled to Philadelphia, New York and Boston to look at their medical society buildings, and eventually, the Philadelphia architectural firm, Ellicott & Emmart was selected to design and build the new Faculty building. Every detail of the building held her imprimatur, from the graceful staircase, to the light-filled reading room, and all of the myriad details of the millwork, marble tesserae, and most of all, the four-story cast iron stacks. She was on-site, climbing up unfinished staircases, checking out the progress of the building, which was built in less than one year at a cost of $90,000.
Among the features of the new building was a fourth-floor apartment for her. She referred to it as the "first penthouse in Baltimore" and it had a garden and rooftop terrace. The library collection eventually grew to more than 65,000 volumes from medical and specialty societies around the world. Journals were traded back and forth, and physicians eagerly anticipated the arrival of each new issue. At the same time, Marcia was involved in the Medical Library Association as one of eight founding members. The MLA promotes medical libraries and the exchange of information. One of the earliest mandates of the MLA was the Exchange, a distribution and trade service for those who had duplicates or little-used books in their collections. Initially, the Exchange was run out of the Philadelphia medical society, but in 1900 it was moved to Baltimore and Marcia oversaw it. Several hundred periodicals and journals were received and sent each month, a huge amount of work for a tiny staff. In 1904, the Faculty had run out of room to manage the Exchange, so it was moved to the Medical Society of the Kings County (Brooklyn). But without Marcia's excellent administrative skills, it floundered and in 1908, the MLA asked Marcia to take charge once again.
In 1909, when the new Faculty building opened, there was enough room to run the Exchange and with the help of MLA Treasurer, noted bibliophile and close friend, Dr. John Ruhräh, it once again became successful. Additionally, Marcia and Dr. Ruhräh combined forces to revive the MLA's bulletin, which had all but ceased publication in 1908, taking the Exchange with it. This duo maintained editorial control from 1911 until 1926. In 1934, around the time of Dr. Ruhräh's death, Marcia became the first “unmedicated” professional to head the MLA. During her tenure, the MLA incorporated, the first seal was adopted, and the annual meeting was held in Baltimore. Marcia wanted to write the history of the MLA once she retired from full-time work at the Faculty, but her health was beginning to fail. She had back problems and had suffered a serious burn on her shoulder as a young woman, possibly from her time running a summer camp, Camp Seyon, for young ladies in the Adirondack Mountains. In 1946, a celebration was planned to honor Marcia's 50 years at the Faculty. But she was adamant that the physicians wait until November, the actual date of her 50 years. However, they knew she was gravely ill, and might not make it until then, so a huge party was held in April. More than 250 physicians attended the celebration, but the ones she was closest to in the early years, were long gone. She was presented with a suitcase, a sum of money to use for travelling, and her favorite painting of Dr. John Philip Smith, a founder of the Medical College in Winchester, Virginia. It was painted by Edward Caledon Smith, a Virginia painter who had been a student of the painter Thomas Sully.[4] She adored this painting and vowed, jokingly, to take it with her wherever she went.
The painting was not to stay with her for very long, for she died in November 1946, and left it to the Faculty in her will. Her funeral was held in the Faculty's Osler Hall, named for her dear friend. More than 60 physicians served as her pallbearers, and she was buried at Baltimore's Green Mount Cemetery. In 1948, the MLA decided to establish an award in the name of Marcia Crocker Noyes. It was for outstanding achievement in medical library field and was to be awarded every two years, or when a truly worthy candidate was submitted. In 2014, the Faculty began giving a bouquet of flowers to the winner of the award in Marcia's name, and in honor of her work. Much evidence exists for this tradition, as we know that the physicians, especially Drs. Osler and Ruhräh, frequently gave her bouquets of flowers. Marcia also cultivated flower gardens at the Faculty and decorated the rooms with her work.
Today, the MedChi building is open for tours and if the rumors are to be believed Ms. Marcia Crocker Noyes is still at work in her beloved library as the "resident ghost" [1][5]
NOTE: This article has been modified from the original Wikipedia article on Marcia Crocker Noyes. The article itself is well-written with interesting images of the subject. I would encourage you to visit it. The second insert is from book 00736 in my personal library and shows in pencil, the incredibly small handwriting of Marsha C. Noyes.
Sources:
1. "Marcia, Marcia, Marcia" MedChi Archives blog.
2. "Marcia C. Noyes, Medical Librarian" (PDF). Bulletin of the Medical Library Association. 35 (1): 108–109. 1947. PMC 194645
3. Smith, Bernie Todd (1974). "Marcia Crocker Noyes, Medical Librarian: The Shaping of a Career" (PDF). Bulletin of the Medical Library Association. 62 (3): 314–324. PMC 198800Freely accessible. PMID 4619344.
4. Edward Caledon BRUCE (1825-1901)"
5. Behind the scenes tour MedChiBuilding
"Clinical Anatomy Associates, Inc., and the contributors of "Medical Terminology Daily" wish to thank all individuals who donate their bodies and tissues for the advancement of education and research”.
Click here for more information
- Details
- Written by: Efrain A. Miranda, Ph.D.
The ganglionated plexi are an important part of the rhythm control system of the heart. One of the problems with the concept of the ganglionated plexuses (also known as ganglionated plexi or GPs) is that these structures are rarely mentioned in medical education and are known only to specialists. People looking for information on this topic rarely find a detailed explanation. This article’s objective is to explain these structures and their function, as well as include some historical vignettes. We will start with a review of the structure and organization of the human nervous system.
The nervous system is a complex network responsible for coordinating bodily functions and maintaining homeostasis, that is, to maintain a constant internal environment despite a variable external environment. It operates as the body’s control center, enabling interaction between different parts of the body and facilitating interactions with the environment, both external and internal. It is composed of two main types of cells: neurons and neuroglia, also known as glial cells. Neurons are responsible for transmitting information through electrical and chemical signals. Glial cells support and protect neurons, also helping in their nutrition.
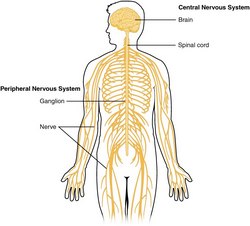
The nervous system is divided into a central nervous system (CNS) and a peripheral nervous system (PNS). The CNS is that portion (brain and spinal cord) contained within the craniospinal cavity and the PNS is composed of neurons and nerves outside this cavity.
It must be noted that these divisions of the nervous system are didactic, that is, they have been created to teach and understand its organization. The fact is that humans have only one nervous system.
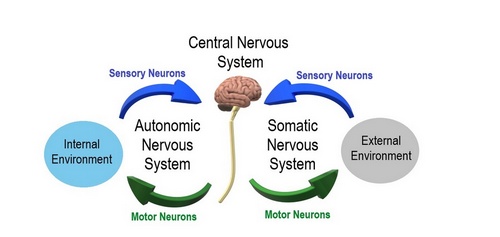
Another division of the nervous system is that of a somatic component and an autonomic component. The somatic component controls voluntary movements and conveys sensory information to the CNS. The autonomic nervous system (ANS) manages involuntary functions such as heartbeat, digestion, urination, etc. For this article we will focus on the ANS.
The ANS is further divided into a sympathetic division, which prepares the body for stress or emergencies (fight or flight response); and a parasympathetic division, which conserves energy, slows down the body and restores the body to a state of calmness (rest and relaxation). Within the ANS we will find motor (efferent) and sensory (afferent) neurons.
Within the PNS and as part of the ANS there are accumulations of neuronal bodies within or close to the internal organs of the body. Each one of these is called a ganglion (Pl. ganglia). The most recognized ganglia are those that compose the sympathetic chain and those related to the origin of most of the branches of the abdominal aorta (Superior mesenteric, celiac, renal, inferior mesenteric, etc.)
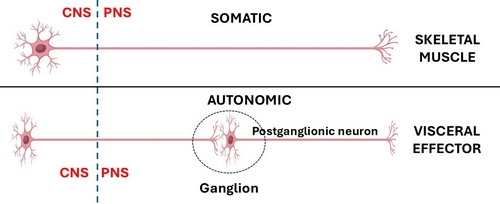
In the case of the CNS somatic (bodily, voluntary) motor organization, is composed of one neuron that is usually located in the frontal lobe motor cortex of the brain in the area known as the precentral gyrus. The distal motor effector of these neurons are distributed to striated (voluntary) muscles throughout the body.
The ANS has been classically described as a 2- neuron system. The body of the first one is found in the CNS and is called the preganglionic neuron, the body of the second one is found in a PNS ganglion and is called the postganglionic neuron, which connects to an autonomic effector (smooth muscle or glands). John Newport Langley (1852 – 1925) a British physiologist coined the terms “preganglionic” and “postganglionic”.
This classic 2-neuron description works for glands and some organs, but it does not consider organs that have rhythmic activity, such as the digestive system and its peristalsis, the heart, ureters, etc.

AI generated image of a neural network
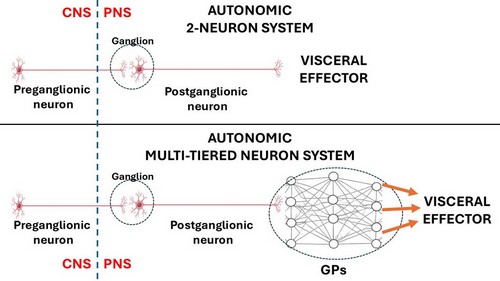
The rhythmicity of these organs is so complex that it requires additional neurons within the muscle layers of each organ. These neurons are found in small intramural ganglia which interconnect with other ganglia by way of nerve fibers forming a complex meshwork (Lat: Plexus). These are the ganglionated plexi (GPs) and their function is to reduce the workload of the CNS by working mostly on their own with oversight of the CNS. These GPs have multiple interconnections forming a neural network, quite similar (on a smaller scale) to the organization of the brain.
Thus, the ANS should be considered as a 2- neuron system in cases such as glands and some organs but, in the case of rhythmic organs there will be ganglionated plexi between the postganglionic neuron and the effector, making it a multiple-tiered system depending on the number of neurons that are interconnected within the GPs.
The two most well-known ganglionated plexi are those found in the muscular and submucosal layers of the small intestine. These plexuses are also found in all the digestive system organs but are more easily studied in the small intestine. They are the plexus of Meissner (submucosal) and the plexus of Auerbach (myenteric). Together they control peristalsis, regulate local secretion and absorption, and modulate blood flow. These nerve plexuses were first described by Leopold Auerbach (1828 -1897) in 1857 and Georg Meissner (1829 – 1905) in 1864.
Because of the importance of these GPs and their activity, they have been called the “enteric nervous system” or the “little brain of the gut” by scientists and journalists. In my opinion, this moniker isolates these GPs from their function as part of the whole of the nervous system.
Another organ that has the same two-layered GPs is the ureter, as described by Pieretti (2018). Neurons (GPs) have been found in the walls of the urethra.
There are organs that have both voluntary and involuntary innervation, such as the urethra, urinary bladder, parts of the anal sphincters, etc. An interesting example of this is the respiratory diaphragm. Most of the day, we are not aware of it working (involuntary – ANS dependent), but if you want to, you can take a deep breath at will (voluntary – somatic dependent)
GPs have been described in the heart, which is a quintessential rhythmic organ. Our life depends on its rhythmicity which helps pump blood through the body. Even before the GPs were discovered, the innervation of the heart was described in detail by Antonio Scarpa (1752 – 1832) in his book “Tabulæ Neurologicæ” published in 1794.
The first to describe GPs in the heart was Robert Remak (1815 – 1865) in 1838, they were known as ”Remak’s ganglia”. Later studies were carried out by Woolard (1926), Singh (1996), Pauza (2000), and others. In these research papers great images of the neuron component of these GPs were published.
Before continuing with the topic of the GPs it is necessary to divert slightly to discuss the conduction system of the heart. The conduction system of heart has been described as a group of nodes and fibers that carry electrical impulses within the heart. (sinoatrial node, interatrial and atrioventricular bundles, atrioventricular node, bundle branches, Purkinje fibers). What is not commonly known is that this system is not formed by neurons and nerves, but by specialized cardiomyocytes (heart muscle cells). Each one of these specialized cells has an automaticity that allows them to beat at a specific number of beats per minute (bpm). The further distally within the conduction system the slower the bpm of each of these muscle cells.
So, if each muscle cell has a predetermined bpm count, how does the heart rhythm increase or decrease when needed? The answer lies in complex reflexes mediated by the GPs and in the interaction of the ANS with the GPs of the heart.
The GPs and their interconnections form the intrinsic cardiac nervous system (ICNS), which has several important functions:
Heart Rate Regulation: The GPs modulate the activity of the sinoatrial (SA) node, which is responsible for initiating the heartbeat.
Conduction Modulation: The GPs also modulate the conduction pathways in the heart, particularly the atrioventricular (AV) node and Purkinje fibers, ensuring coordinated contractions of the heart muscle.
Cardioprotection: The GPs help protect the heart by altering cardiac output in response to exercise, stress, ischemia, etc., maintaining heart function and helping homeostasis under challenging conditions.
Coordinate Local Reflexes: The GPs integrate sensory information from the heart and coordinate local reflexes, allowing for rapid adjustments to changes in the heart's environment.
These functions highlight the importance of ganglionated plexuses in maintaining heart health and responding to physiological demands. Dysfunction in these plexuses can lead to various cardiac issues, including arrhythmias, particularly atrial fibrillation (AFib), and heart failure.

J. Andrew Armour
Cardiac GPs are found in the cardiac wall, although most of them are found intramural and superficial (epicardially) (Tan, 2006), and also embedded in epicardial fat, in the areas devoid of pericardium (formed by the pericardial reflections) around the pulmonary veins, superior vena cava and around the ligament of Marshall. Although GPs are found in the atria and ventricles, there are 5 times more atrial than ventricular GPs (Armour, 1997).
In the study of the GPs, J. Andrew Armour MD, PhD, has been a foundational figure in neurocardiology, and his work has shaped our modern understanding of the intrinsic cardiac nervous system (ICNS), and the GPs. He proposed that the ICNS is not just a passive relay for ANS signals, but a complex integrative network of efferent (motor) neurons, afferent (sensory) neurons, and local circuit neurons that form the GPs. Through his work, Armour argued that the heart’s neuronal network is sufficiently rich to process information, participate in local reflexes, and modulate cardiac function independent of central nervous system input. This can be proven by the heart working normally in heart transplants, completely separated from the recipient's nervous system.
In conclusion, there are two points to consider:
1. The 2-neuron organization of the ANS must be expanded to consider all organs that have rhythmicity and have GPs forming an intramural neural network, following this schema.
2. Our understanding of the rhythmicity of the heart has been one-sided, focusing only on the cardiomyocyte-based conduction system of the heart. This leads to surgical and minimally invasive treatments that do not consider the ICNS and GPs, as well as the location of most of the GPs on the external layers of the heart. A unified approach to these structures will certainly help those patients with cardiac arrhythmias and their surgical treatment.
Sources:
1. Stavrakis S, Po SS. “Ganglionated Plexi Ablation: Physiology and Clinical Concepts.” Heart Rhythm 2017. Arrhythm Electrophysiol Rev 6(4):186–190
2. “The Intrinsic Cardiac Nervous System and Its Role in Cardiac Function” Fedele L, et al. 2020. J Cardiovasc Dev Dis 7(4):54.
3. "A brain within the heart: A review on the intracardiac nervous system" Campos, I.D.; Pinto, V.J Mol and Cell Cardiol. 2018 119:1-9
4. "Anatomical Distribution of Ectopy-Triggering Plexuses in Patients with Atrial Fibrillation" Kim, MY et al Circulation" Arrythmia and Electrophysiol 2020. 13:9
5. “Morphology, Distribution, and Variability of the Epicardiac Neural Ganglionated Subplexuses in the Human Heart” Pauza, D., Skripka, V et al The Anatomical Record 259:353–382 (2000)
6. “Gross and Microscopic Anatomy of the Human Intrinsic Cardiac Nervous System” Armour JA et al, (1997) The Anatomical Record 247:289-298
7. “The Junction Between the Left Atrium and the Pulmonary Veins, An Anatomic Study of Human Hearts” Nathan, H; Eliakim, M. Circulation (1966) 34: 412-422
8. “Autonomic innervation and segmental muscular disconnections at the human pulmonary vein-atrial junction: implications for catheter ablation of atrial-pulmonary vein junction” Tan AY, Li H, Wachsmann-Hogiu S, Chen LS, et al J Am Coll Cardiol 2006;48:132–143
9. “Topography of cardiac ganglia in the adult human heart” Singh S, Johnson PI, Lee RE, Orfei E, Lonchyna VA, Sullivan HJ, et al. J Thorac Cardiovasc Surg 1996; 112:943-53
10. Woollard, H. H. “The Innervation of the Heart.” Journal of anatomy (1926) 60 Pt 4: 345-73. 4
11 “Tabulæ Neurologicæ ad illustrandam historiam anatomicam cardiacorum nervorum, noni nervorum cerebri, glossopharyngæi, et pharyngæi ex octavo cerebri. ” Scarpa, A. 1794 Balthasarem Comini Pub.
12.“The Involuntary Nervous System” Gaskell, W. H.2nd Ed. Longman, Green & Co. 1920
13. “Cardiac anatomy pertinent to the catheter and surgical treatment of atrial fibrillation”. Cox. JL et al J Cardiovasc Electrophysiol 2020 Aug;31(8):2118-2127. doi: 10.1111/jce.14440.
14. "Minimally Invasive Surgical Treatment of Atrial Fibrillation: A New Look at an Old Problem" Randall K. Wolf, Efrain A. Miranda, Operative Techniques in Thoracic and Cardiovascular Surgery, 2024,
15. “Correlative Anatomy for the Electrophysiologist: Ablation for Atrial Fibrillation. Part I: Pulmonary Vein Ostia, Superior Vena Cava, Vein of Marshall” Macedo PG, Kapa S, Mears JA, Fratianni A, Asirvatham SJ. J Cardiovasc Electrophysiol. 2010 Jun 1;21(6):721-30.
16. “Treatment of lone atrial fibrillation: minimally invasive pulmonary vein isolation, partial cardiac denervation and excision of the left atrial appendage” Wolf, R.K. Ann Cardiothoracic Surg. 2014; 3:98-104
17. “Neurocardiology: Anatomical and Functional Principles” Armour, J.A. Institute of HeartMath, Boulder Creek, CA, 2003
18. “The enteric nervous system: a little brain in the gut" Annahazi, A. ∙ Schemann, M. Neuroforum. 2020; 26:31-42
19. “The Little Brain Inside the Heart” Dr. José Manuel Revuelta Soba Professor of Surgery. Professor Emeritus, University of Cantabria, Spain
Images
Image of the CNS - Public Domain. Source: WikipediaCreative CommonsAttribution 4.0 International license.
Autonomic and Somatic Image. Public Domain by Christine Miller. Source: WikipediaCreative CommonsAttribution 4.0 International license.
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 1429
During the last year the number of artificial intelligence (AI) anatomical and surgical images found in websites and social media (Facebook, Instagram, TikTok, etc.) has grown exponentially. The same has happened with posts and articles from so-called experts targeting the public, medical students, and health professionals. Unfortunately, many of these have glaring anatomical errors (1). The reason for this trend is an unscrupulous race to obtain an increased number of followers which leads to monetization of a website, posts, individuals, or groups.
The authors using AI prompts that create these images publish them as fast as they are produced without a thought as to the correctness of what’s being created by AI. Their followers, also without a thought or question of the content or image, like, share, copy and distribute these on the Internet. An example is the publication (now retracted) of rat stem cells using AI imagery. The image generated is of such bad taste, yet it was peer-reviewed and published. The image shows a rat with large genitalia. I cannot in good conscience publish the image here. For more information see “Sources” 2, 3 (a newspaper article on the problem), and the publisher's retraction.
Another glaring example is this video that shows the heart valves in action. It is not accurate. The movement and synchrony of the valves is wrong, they do not open, the pulmonary valve, which normally has three leaflets, shows four. The coronary arteries are in the wrong location and there an extra coronary on the right side. No one could live with that aortic valve. Yet, this image has 10.K likes and 2.5K shares! Ignorance is being shared. Just because it looks sexy, it does not mean that it is correct! (1).
The problem is that these websites and images are being used by health care professionals, students, and medical industry training groups without questioning the accuracy of the information being copied and redistributed externally and internally in training documents. This is not an uninformed statement. I have seen it personally, not in one, but in several companies.
The pressure that training groups in the medical arena and in other industries have is to reduce costs and training time. For those of you who know me and whom I have trained, you may recall how two decades ago training for a surgical devices representative could take six, ten weeks, and sometimes longer. I do know of one company that required a six-month internship before considering someone ready! Many of those who underwent this training are today in high-level corporate positions or by now have retired very well.
Today, the race is on to reduce face-to-face training time because it is perceived to be expensive, but the cost of poor and inaccurate training for a company is much, much more expensive! Other trends are the use of computer-based training (reduces cost), and reducing the amount of information passed on to the trainee (reduces time). The interesting situation here is that there are forces within these companies that are uninterested in learning more, just training enough to do a basic job.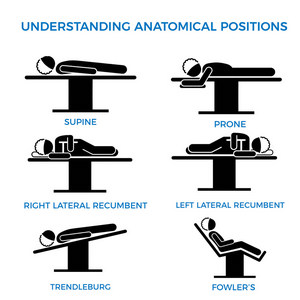
Another alarming trend is that knowledge acquired in training is geared toward acing the internal tests (checking the boxes) and not necessarily geared toward working use of that knowledge when interacting with a medical professional.
The need to generate training material forces many to copy and paste images and concepts from the Internet. Just because a concept is available on the Internet does not make it correct. In fact, there are many reputable websites and books that have erroneous information. Here is an example from a medical devices company that shows “anatomical positions” (there is only one anatomical position) when the image should be labeled “surgical positions”. If you click on the image you will see a larger image with corrections.
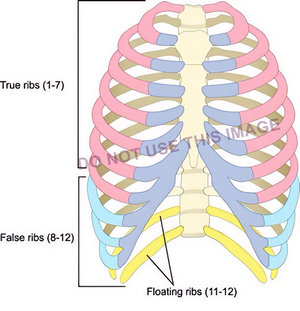
The next image by a reputable medical illustrator shows the groups of ribs, but there is a mistake: False ribs are only 8 – 10, not 8-12! Yet this image is being used for training. I do not know if this image was incorrectly labeled by a third party.
I have seen many companies give a sales representative, manager, engineer, or other employee the responsibility to develop presentations and computer-based training, and what do they do? Go on the Internet for information. Here is where that computer acronym becomes a painful reality: GIGO “garbage in, garbage out”. For me, this is unacceptable, as this could affect a patient!
The only way to ensure the information is correct is to use an expert (but of course that’s not free). This leads to another problem: The Dunning-Kruger effect. This is a phenomenon where some people believe that they are much more competent, knowledgeable, or capable than they really are. Furthermore, they convince their peers and their company of it. David Dunning and Justin Kruger In their 1999 article coined the term “unskilled and unaware of it”, which is sadly becoming commonplace today.
Another example of these AI generated images is this view of the spine, spinal cord and spinal nerves and branches. The zygapophysial joints look fused (they are not), the dural (thecal) sac fills the vertebral canal (it does not), the spinal nerve and its branches are wrong. Interestingly, I did comment on the mistakes in the image to the author. The answer I received was: ”the more easy the illustration, the more the students will get into it. A complicated high info video may scare undergraduate freshers. That's why I post things in simple”. So, is it correct to teach something wrong? Call me old-fashioned, but I do not think so.
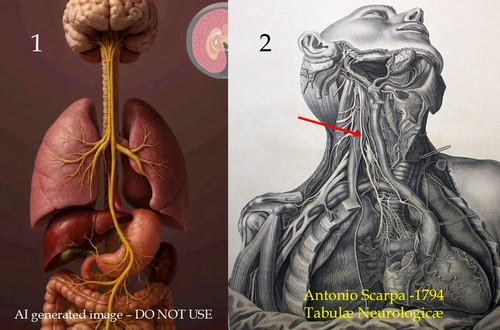
I could go on and on with these examples. Case in point: Image 1 is an AI generated image of the vagus nerve. It is wrong on so many levels! Contrast that with image 2, which is a public domain image from the 1794 “Tabulae Neurologicae” by Antonio Scarpa. The arrow shows the right vagus nerve.
Text available online should not be copied without ensuring that it is correct. An example: “The lungs are enclosed by the pleural sacs, which are attached to the mediastinum”. The first part is correct, each lung is contained in a separate pleural sac (only 86% of the time, the rest of us may have a communication between both pleural sacs). But the pleural sacs are not attached to the mediastinum! The mediastinum is a concept, the region between the pleural sacs, not a real structure.
Lastly, there are copyright issues where it has become so simple to highlight, copy, and paste that many do not think twice about using proprietary text and images without considering the consequences of this activity. One of the most used and abused books is Frank Netter’s Atlas of Human Anatomy. You can see it all over the Internet in posts that appear daily. This is a technique to increase traffic and clicks. Just because it was published in one of these social media posts, does not mean that we can freely use it in training materials. This abuse is rampant in social media where so-called experts copy and paste images from books and other websites.
Additionally, these deepfake "experts" are generating click-bait videos that use audio tracks (probably also AI generated from text) scaring and fooling people on social media.
The Internet is an extremely powerful tool, yet it is important to remember that not everything on the Internet is true, accurate, and readily available for copy and paste functions. Not all information should be believed at face value. I sincerely hope that this trend changes.
Why do I think this is so important? In the medical devices industry our first responsibility is to the patient and to be able to provide the best care. A healthy distrust of every bit of information we use, and investing time and resources to attain accuracy will help us toward this objective.
Following are some additional examples, some so fake as to be hilarious, but someone is watching them and believing what they are seeing and hearing.
The first video shows structures inferior to the transverse colon that do not exist, besides that, peristaltic movements are not like that... it looks more like heart contractions! The second video shows a moving thyroid gland and the vascular and nerve structures are all wrong! The third one is ridiculously wrong!! These videos hare captioned in Spanish, but you can find the same in any other language.
Personal note: I have purposely tried to avoid identifying individuals, websites, companies, etc. while writing this article. I have also decided to add this AI generated image to my list of pet peeves.
"The whole problem with the world is that fools and fanatics are always so certain of themselves, and wiser people so full of doubts.”
Bertrand Russel
Sources:
1. “It looks sexy, but it is wrong. Tensions in creativity and accuracy using genAi for biomedical visualization” Zimman, R; Saharan, S; McGill, G; Garrison, L. 2025 https://arxiv.org/pdf/2507.14494
2. “RETRACTED “Cellular functions of spermatogonial stem cells in relation to JAK/STAT signaling pathway” Guo,X; Dong, L; Hao, D. Frontiers in Cell and Developmental Biology. 2024 PDF Link here
3. “AI-generated nonsense about rat with giant penis published by leading scientific journal” The Telegraph, 2024.
4. “Emotionally unskilled, unaware, and uninterested in learning more: Reactions to feedback about deficits in emotional intelligence” Sheldon, O. J., Dunning, D., & Ames, D. R. (2014). Journal of Applied Psychology, 99(1), 125–137. https://doi.org/10.1037/a0034138
5. “Unskilled and unaware of it: How difficulties in recognizing one's own incompetence lead to inflated self-assessments” Kruger, J., & Dunning, D. (1999). Journal of Personality and Social Psychology, 77(6), 1121–1134. https://doi.org/10.1037/0022-3514.77.6.1121
6. “Chapter 5- The Dunning–Kruger Effect: On Being Ignorant of One's Own Ignorance” Olson, HM; Zanna, MP. Advances in Experimental Social Psychology (2011) – 44: 247-296 https://doi.org/10.1016/B978-0-12-385522-0.00005-6 (these are snippets, not the whole chapter)
7. "How people are being tricked by deepfake doctor videos on social media" New York Post July 17, 2024
8. "Positioning in Anesthesia and Surgery" Martin, JT; Warner, MA 3rd Ed. 1997 USA W.B. Saunders
9. https://elearningdoc.com/the-hidden-costs-of-inadequate-training/
- Details

In the many years I have lectured for the medical industry, the one word that representatives, managers, engineers, etc. usually do not master is “morbidity”.
In most cases when I ask for a definition of “morbidity” most in the audience try to guess and in the medical arena and medical devices industry guessing is not acceptable. So, I would like to ask those who follow my articles and posts to repost this information so that it is available for as many people as possible. By the way, the most common answer I receive is that "morbidity" means death...not so!!
The term “morbidity” arises from the Latin word morbus which means Ill, sick, or sickly. It has been in use in English for centuries and what today we call “pathology” used to be known as “morbid anatomy”. There are many terms in medical history that included the term “morbid” such as “morbus indecens” (venereal diseases); morbus gallicus (syphilis), “morbus sacer” (epilepsy, the sacred disease), etc.
To be simple, today we use the term “morbidity “to mean “complications”, As an example, when we use the term “postoperative morbidity” what we’re trying to say is simply “postoperative complications”. When you ask what is the morbidity associated with a certain treatment (a new medication, as an example) what we’re trying to say is what are the complications associated with that medication or treatment.
Now, the associated term “comorbidity” causes the same problems. Many think they know its meaning, but they are actually guessing, and it can lead to embarrassing conversations between a medical device representative and a surgeon or physician.
The term comorbidity is a portmanteau (French) word, that is, a word formed by the mixing of parts of two words. In this case, the components are COncommitant (meaning associated) and MORBIDITY, (meaning complications). Simple stated, comorbidity means “associated complications”. As a side note, do not add a hyphen, as in co-morbidity: this is wrong.
There are many portmanteau words in English, such as “brainiac”; “bromance”, “Brexit”, “fog”, etc. For additional portmanteau non-medical words follow this link.
Sources.
1. "The origin of Medical Terms" Skinner, AH 1970
2. "Medical Meanings - A glossary of Word Origins" Haubrich, WS. 1997
- Details
- Written by: Efrain A. Miranda, Ph.D.
- Hits: 6937
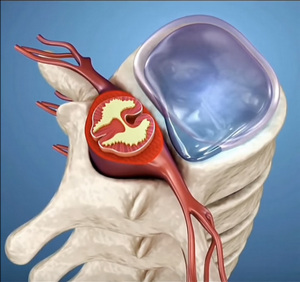
The persistent left superior vena cava (PLSVC) is the most common anatomical variation or anomaly in thoracic anatomy. It is present in 0 .3 to 0 .5% of the general population, but it can be present in 5% to 10% of patients who present some type of congenital cardiac malformation.
The most common presentation of this anomaly is where the PLSVC shares the venous drainage with a normal right superior vena cava. In other cases, the PLSVC is present, but there is total absence of the right superior vena cava. In these cases, the condition can be asymptomatic and can be discovered intra or preoperatively. The PLSVC is shown with a yellow arrow in the accompanying image.
In some cases, the PLSVC opens into the left atrium, causing a right to left cardiac shunt, a condition that is clearly symptomatic.
Embryologically, the development of the (right) superior vena cava begins with a similar counterpart on the left side of the embryo, the left anterior cardinal vein.
The left cardiac horn and left anterior cardinal vein eventually form the coronary sinus while its superior portion obliterates, becomes non- patent and forms the duct of Cuvier and the ligament of Marshall. A portion of the left anterior cardinal vein remains patent and forms the oblique vein of the left atrium (also known as the vein of Marshall). The vein of Marshall is found at the base of the left atrial appendage.
When present, the diameter of the PLSVC is usually quite larger than the average diameter of a normal coronary sinus, and because of the increased flow into the right atrium, the valves of Thebesius (valve at the ostium of the coronary sinus) and the valve of Vieussens (valve found at the end of the great cardiac vein and the start of the normal coronary sinus) are either absent or present with substantial reduction in size.
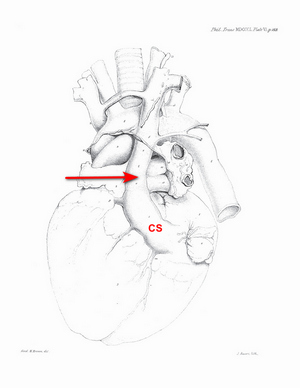
The history of the discovery and description of the PLSVC is not clear. There are many attributions, but what is undeniable is that the first complete and detailed description of this anatomical variation was done by John Marshall in 1850. The following image shows the original drawing (Plate VI) in his article “On the Development of the Great Anterior Veins Man and Mammalia; including an Account of certain remnants of Fœtal Structure found in the Adult, a Comparative View of these Great Veins the different and an Analysis of their occasional peculiarities in the Human Subject”.
While often clinically silent, PLSVC has important implications for central venous access, pacemaker lead placement, and cardiac surgery.
Personal note: My personal thanks to my good friend and contributor to "Medical Terminology Daily", Dr. Randall K. Wolf for the surgical image.
The following YouTube video by Medical Snippet with animations and drawings by Karthik Easvur provides a detailed description of the formation of the superior vena cava and the PLSVC.
Sources:
1. “Persistent left superior vena cava”. Tyrak KW, Holda J, Holda MK, Koziej M, Piatek K, Klimek-Piotrowska W. Cardiovasc J Afr. 2017 May 23;28(3):e1-e4. doi: 10.5830/CVJA-2016-084. PMID: 28759082; PMCID: PMC5558145.
2. “Persistent left superior vena cava: review of the literature, clinical implications, and relevance of alterations in thoracic central venous anatomy as pertaining to the general principles of central venous access device placement and venography in cancer patients.” Povoski SP, Khabiri H. World J Surg Oncol. 2011 Dec 28;9:173. doi: 10.1186/1477-7819-9-173. PMID: 22204758; PMCID: PMC3266648.
3. “Absent Right Superior Vena Cava and Persistent Left Superior Vena Cava: An Incidental Finding.” Joshi, Swati, and Ajmer Singh. Annals of Cardiac Anaesthesia, 2nd ed., vol. 26, no. 4, 2023, pp. 433–34, https://doi.org/10.4103/aca.aca_91_23.
4. "Absent right superior vena cava in visceroatrial situs solitus.” Bartram, U., Van Praagh, S., Levine, J. C., Hines, M., Bensky, A. S., & Van Praagh, R. 1983 Am J Card, 52(10), 1262–1268.
5. “Superior vena caval abnormalities: their occurrence rate, associated cardiac abnormalities and angiographic classification in 542 patients” Buirski, G., Jordan, S. C., Joffe, H. S., & Wilde, P. (1986). Cardiovasc Interv Rad, 9(6), 357–362.
6. "Persistent Left Superior Vena Cava with Absent Right Superior Vena Cava" Pate, Y; Gupta, R. Methodist DeBakey Cardiovasc J. 2018. 14:3. 2223-235. DOI: 10.14797/mdcj-14-3-232
7. “On the Development of the Great Anterior Veins Man and Mammalia; including an Account of certain remnants of Fœtal Structure found in the Adult, a Comparative View of these Great Veins the different and an Analysis of their occasional peculiarities in the Human Subject” 1850 Phil Trans R Soc 140:133 - 170. To download this article click here.
Video courtesy of Medical Snippet. The video, animations and drawings are the property of their owners. We encourage viewers to follow and subscribe to their respective YouTube channels.
Image of Marshall's Plate VI modified from the original. Public domain.
- Details
- Written by: Efrain A. Miranda, Ph.D.
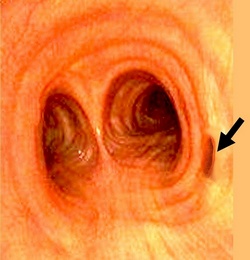
Tracheal bronchus (pig bronchus)
After writing an article on the tracheal bronchus, I was asked to describe the anatomy of the pig (Sus scrofa domesticus) lung and a comparison with the human lung. The function and general structure of the pig lung is similar to the human. Anatomy-wise... there are several differences.
It is important to understand that directional terminology used to describe the anatomy of the pig is different than that used for the human. We use the anatomical position for humans, but as the pig is a quadruped, there is no such position for the pig, and veterinary terminology is used.. For further information on this topic, click here.
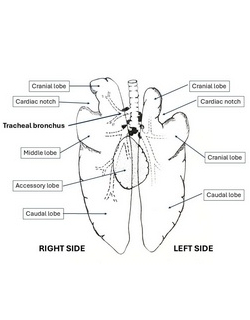
Following are two images. The first one is an anterior view of the human lungs and their tracheobronchial tree. The second image shows a ventral view of the pig tracheobronchial tree and both lungs.
The first difference with the human lung is that there are two cardiac notches, right and left (the left cardiac notch is larger) whereas the human has only one, on the left lung.
The structure of the tracheobronchial tree in both species is similar, with incomplete cartilaginous rings and a posterior (dorsal) membrane that closes both the trachea and bronchi. Similarly, as the bronchial tree is more distal, the cartilaginous rings break up.
The trachea in the human ranges between 10 to 12 centimeters, while in the pig the trachea is longer, between 25 to 30 centimeters. Both bifurcate at the carina into a right and left main stem bronchus. In both species there is a large number of lymphatic nodes at the tracheal bifurcation (carinal nodes) which drain the lungs.
In the pig, the bronchus for the right cranial lobe arises directly from the trachea, and is known as the "tracheal bronchus". This does not normally happen in the human, and when it does it is considered an anatomical variation called a "pig bronchus", "bronchus suis", or "tracheal bronchus", and it can cause serious problems during intubation in surgery.
In the human, there are normally three lobes on the right side (upper, middle, and lower), and two on the left side (upper and lower). Each lobe has its separate lobar bronchus.
The right lung in the pig has 4 lobes: Cranial, middle, caudal, and an accessory lobe that is ventral and is located in the midline. Each lobe has its own separate lobar bronchus.
The left lung in the pig has two lobes: Cranial and caudal. The left cardiac notch splits the cranial lobe in two segments, but since these segments arise from a common bronchus, they are considered one lobe.
Sources:
1. "Essentials of Pig Anatomy" Sack, W.O.; Horowitz, A. 1982 Veterninary Textbooks, Ithaca, New York..
2. “Bronchial tree, lobular division and blood vessels of the pig lung” Nakakuki, S. J Vet Med Sci. 1994 Aug;56(4):685-9.
3. “Bronchial anatomy and single-lung ventilation in the pig” Muton, WG. Can J Anesth 1999 46:7 p701-703
- Human tracheal bronchus endoscopic image modified from the original. Public domain.
- Anterior view of the human lungs and tracheobronchial tree image by . Patrick J. Lynch, medical illustrator, CC BY 2.5 <https://creativecommons.org/licenses/by/2.5>, via Wikimedia Commons.
- Ventral view of the Ventral view of the pig lungs and tracheobronchial tree image by Dr. Miranda, modified from the original. Public domain.
- Details
- Written by: Efrain A. Miranda, Ph.D.

Tracheal bronchus (pig bronchus)
The tracheal bronchus is a known anatomical variation first described in 1785 by Eduard Sandifort (1742 – 1814), a Dutch physician and anatomist. Sandifort was the first to describe in detail what we know today as the Tetralogy of Fallot, although Nicolaus Steno (1638-1686) mentions the components of this pathology, he did not investigate it in detail as Sandifort did.
It is mostly known as the “pig bronchus” because of its similarity with pig anatomy; it is also called “bronchus suis”. When present, the tracheal bronchus usually arises on the right side of the trachea, about 2 cm. superior to the tracheal bifurcation (carina). The location of the bronchial aperture can vary between the cricoid cartilage of the larynx superiorly, and the tracheal bifurcation inferiorly.
The tracheal bronchus is usually the only airway supplying the upper right lobe of the lung, although it can share the airway with a smaller right upper lobe bronchus. In this case the tracheal bronchus is called “accessory tracheal bronchus”.
In cases where the origin of the tracheal bronchus is higher on the trachea, the trachea may present with distal stenosis. In rare cases, the tracheal bronchus may be found on the left side supplying air to the superior portion of the left lung.
The incidence of a tracheal bronchus varies between 1 to 5%, where the prevalence on the right side is 0.1 to 2% and 0.3 to 1% on the left side. It is sometimes found associated with other congenital abnormalities.
It is usually asymptomatic, but it can be found related to recurrent pulmonary infections, bronchiectasis, chronic bronchitis, and partial airway obstruction. It is most often discovered incidentally, usually during the intubation process for thoracic surgery.
Personal note: My thanks to Dr. Randall K. Wolf for suggesting this article.
Sources:
1. “Observationes anatomico-pathologicae” Sandifort, E. (1785). Lugduni Batavorum: Apud S. & J. Luchtmans.
2. “Congenital bronchial abnormalities revisited” Ghaye, B., Szapiro, D., Fanchamps, J. M., & Dondelinger, R. F. (2001). Radiographics, 21(1), 105–119.
3. “Tracheal bronchus: A rare cause of right upper lobe collapse” Ngernchuklin, P., Sumanac, K., & Behrsin, J. (2006).. Canadian Journal of Anesthesia, 53(12), 1227–1230
4. “Tracheal bronchus” radiopedia.org. Elfeky, M. 2023.
5: ”Bronchus suis – Case presentation” radiopedia.org. Ian Bickle.
Image modified from the original, public domain.